Centralize, Track, and Empower: Facilitating Good Clinical Data Review in Your Clinical Trial
Discover how Revvity Signals helps centralize, track, and empower good clinical data for efficient and compliant clinical trial operations. Learn more here.
Constant changes in clinical trials demands a rapidly adaptive analytic tool.
Adaptive analytics is crucial to the development of a new pharmaceutical product. Decentralized trials and/or new trial designs are causing a rapid increase in the variety of data sources used in trials. These data must be carefully collected and analyzed to meet a growing list of guidelines for Good Clinical Practice (GCP), including International Council for Harmonization of Technical Requirements for Pharmaceuticals for Human Use (ICH) E6(R2) and the impending ICH E6(R3).
To keep up with these changes, drug developers need robust computational and analytical tools to manage clinical trial data and extract the most relevant insight. Beyond using analytics to identify safety, operational, or data issues, collaboration across clinical development teams is essential. As such, your analytic tool should provide integrated collaboration capabilities.
MANAGING CLINICAL DATA COMPLEXITY
Increasingly complex data gathered in decentralized and other new trial designs form much of the evolution in biomedicine. Instead of serving only as end points in clinical trials, data can now be used as inputs for translational research and othe r processes. An effective clinical analytics platform takes into account all the relevant data by:
- Data source connectivity with automated refreshes
- Aggregating and standardizing clinical data
- Effective collaboration around the data
- Empowering the results through adaptive tools, reporting and metrics
When implemented, these four criteria improve a company’s control of its clinical trial data and increase volume of available data. With an evolving data source landscape capable of collecting thousands of data points per second, the challenge is bringing together extensive amounts of information to ensure safety and efficacy. Thus, it's critical to have analytics tools that are data collection agnostic and can adapt to the broadness, depth, and quality of data from collection sources like eDC, ePRO, eCOA, wearables, IXRS, CTMS, central labs, and more.
This reveals a fundamental shift in clinical trials as they move to new models as well as the importance of informatics: to leverage data to derive insights from them and improve human organizational performance. In other words, make the data work for you, not the other way around.
Recently, the Society for Clinical Data Management asked companies about the number of data sources that they now manage and expect to manage three years into the future in clinical studies. As shown in
Figure 1, the number of data sources in clinical trials keeps increasing.
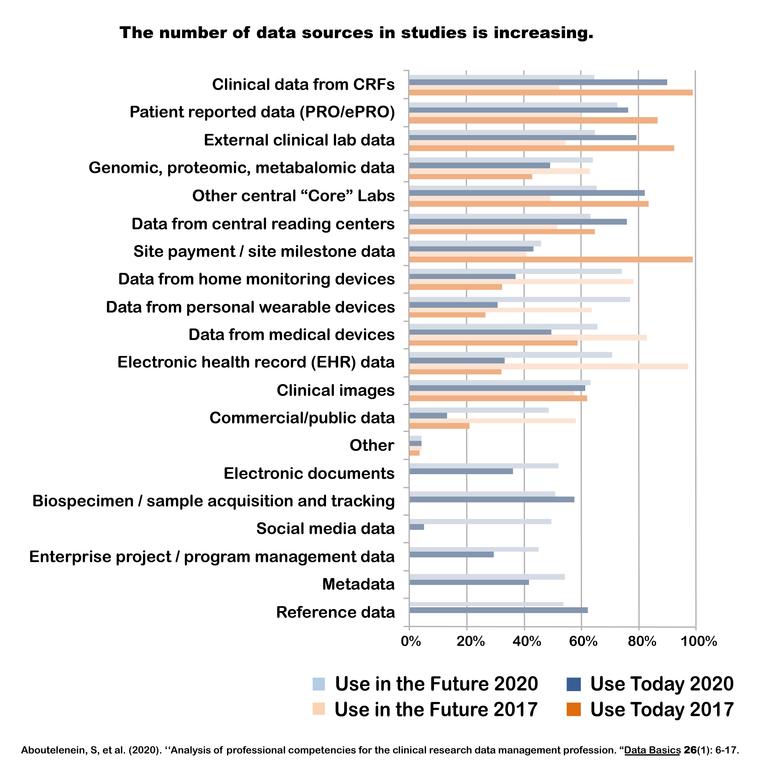
Figure 1. Data sources in studies: 2020 vs 2017.
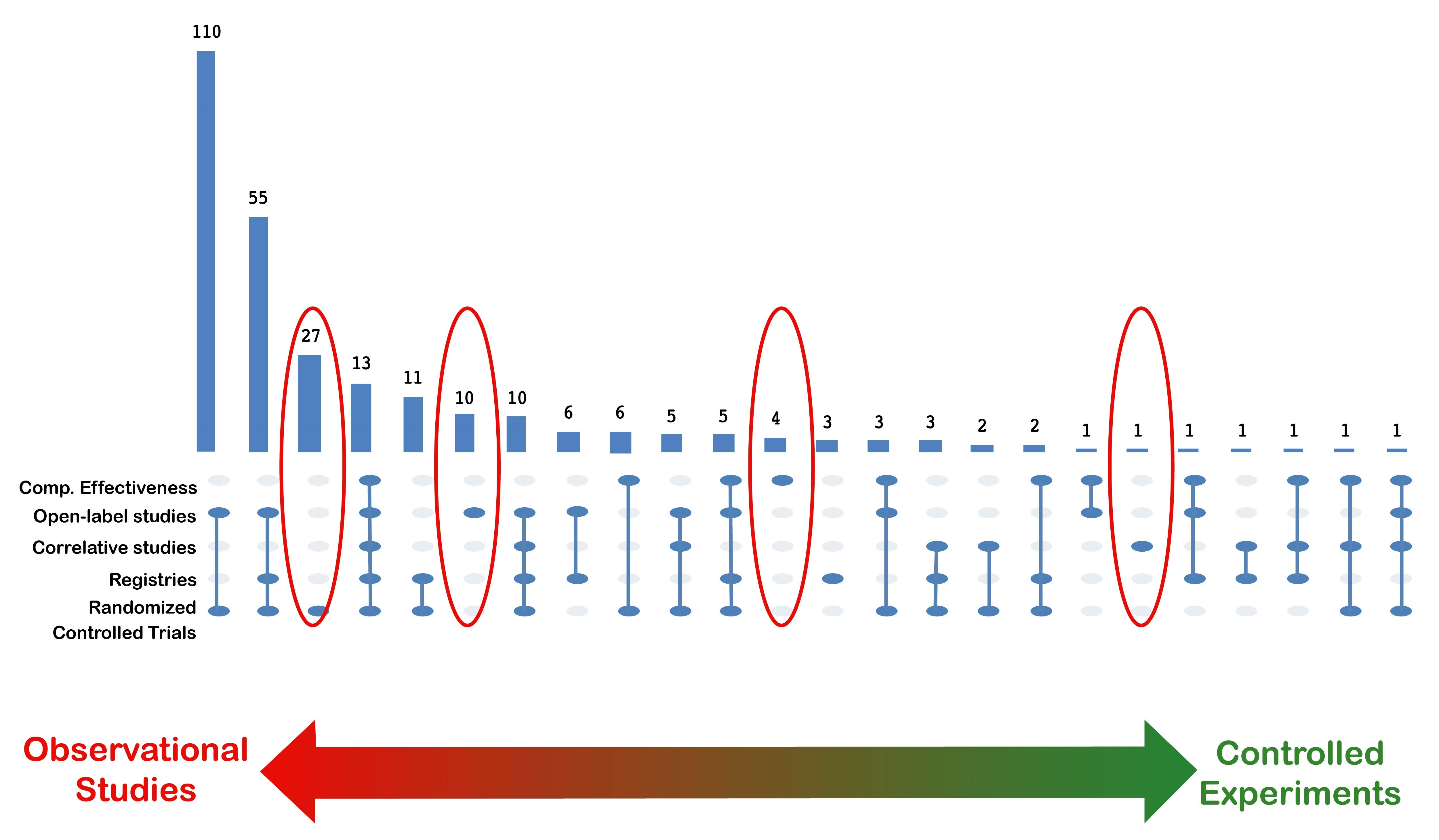 Figure 2. Very few respondents reported supporting only one type of study.
Figure 2. Very few respondents reported supporting only one type of study.
Additionally, a small number of people contacted for this analysis worked on only one type of study, which is indicated by the red ovals in Figure 2. If the respondents noted working on observational studies and controlled experiments. Observational studies use existing data, which differs from real-world data collected electronically in prospective studies. For instance, real-world data are being used for a new or secondary purpose. This leaves no control over real-world data, except to assess its quality, but scientists have some control over prospectively collected data.
Beyond these two basic applications of information, the biopharma industry faces more expectations than ever about how it uses data, especially to improve overall performance and therapeutic development. For one thing, the data are expected to produce an increasingly realistic picture of the natural world. That depends on a rich dataset that can be traced during processing, including keeping track of data manipulations, quality and uncertainty. Today’s information technology helps companies meet these data expectations in four fundamental ways: connectivity, automation, decision support, and deriving new insight from data.
The industry expects these tools to be used throughout future studies, but it will take some time to get there. In a poll of the participants of this webcast of their…
“Most pressing clinical data management challenges where you believe software solutions can best aid,” the top response was
“Data automation and timeliness to get the right data to the study teams
at the right time,”
and many participants also noted challenges in connectivity, visualizations and so on. These answers likely arise from the expanding sources, acquisition speed and volume of data, coupled with the expectations of combining that information so that it can be used as quickly and effectively as possible.
BUILDING A SOFTWARE FAMILY
Revvity Signals started with ELN solutions, and then expanded into suites of scientific software, developed in partnership with Spotfire™. Although these software solutions cover a range of applications, they all focus on one fundamental objective: providing smarter science,
as shown in Figure 3.
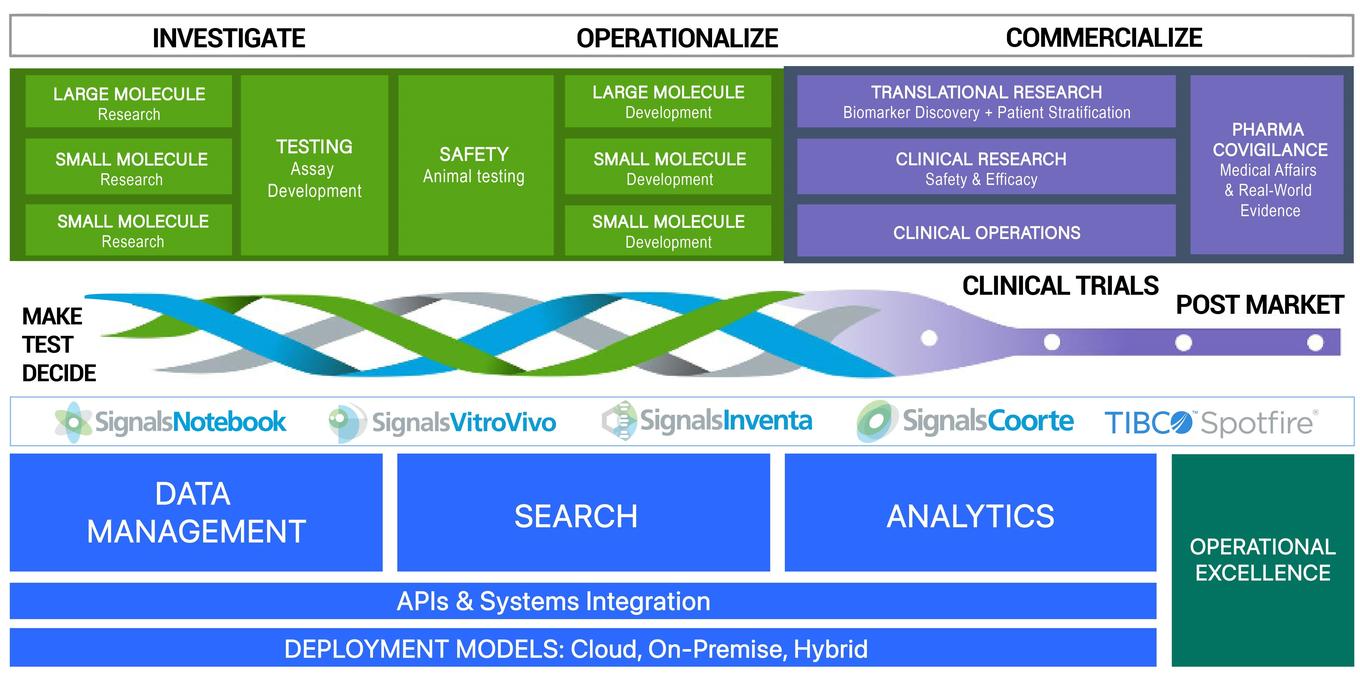
Figure 3. Smarter science.
The solutions from Revvity Signals apply to basic research through post- marketing needs, as well as every step in between. Plus, these tools connect all of the information from one stage to another. This connectivity provides data communication that gives an organization more power to optimize its performance as an organization and as a creator of better therapeutics. This communication takes place between and within departments in a company, but it must also extend to partners working on research and clinical data around the world.
In working with its partners, Revvity Signals finds that most of the industry lacks centralized analysis and communications.
In the best-case scenario, clinical trial analysis and communications are located side-by-side. The increasing diversity of clinical trial types, which run over different lengths, adds to the complex collection and ultimate comprehension of the different kinds of data. Also, this information must be readily accessible across a company. These needs put crucial pressure on organization’s communications capabilities.
Many of today’s companies face a variety of data challenges, sometimes as simple as even getting access to the right data to the right people to make relevant insights. Subsequently, the company wants access as quickly as possible, as well as the ability to query data for specific metrics or to identify trends in the data.
At a fundamental level, an organization needs methods that allow it to answer vital questions. Figure 4 shows that important questions come from across all components of a company’s clinical development operations as an example. From looking for the most recent data to following GCP guidelines, a lack of centralized analysis and collaboration creates a bottleneck at best.

Figure 4. Top questions from our clinical trial partners.
Many biopharma companies face four data challenges. First, a company needs the right data at the right time, which depends on direct connectivity and automation. The second challenge involves making the right data available to all team members by having it properly distributed. This requires aggregation and the ability to reveal relationships across data sets. Third, as clinical trials evolve, a company will deal with new data that must be analyzed in new ways, which will not be possible with inflexible tools. Conversely, modular and adaptive tools can handle changes in circumstances, new kinds of data, or changes in protocols. And lastly, the fourth challenge involves ensuring that a company reviews data with integrated collaboration; only then can a company feel confident that it is controlling its data. This confidence allows a company to act on the data in ways such as simplifying a data audit or data-based decision making.
INTEGRATED SOFTWARE SOLUTIONS
Revvity Signals offers a collection of modules that resolve a series of concerns— depicted in Figure 5 for investigational products for solid-tumor oncology, but applicable to any therapeutic area. First, the Clinical Data Review module provides standard methods of analyzing safety and efficacy. The Clinical Operations module provides data- driven insights to optimize site performance while improving patient safety. Risk-Based Monitoring provides timely information and actionable insights that enable optimal study start-up, budget, and milestone tracking.
The value of centralized communications emerges in the Signals Line Listing Review module, which can be used to manage information that’s been reviewed or tagged for data management querying or for further analysis. In short, this allows personnel to track the status of data and collaborate. The Revvity Signals line-listing review capability is directly integrated within Spotfire, obviating the need for utilizing external tracking tools. Therapy-related modules can also be added to optimize an analysis package (such as RECIST visuals for oncology). The software allows connectivity across a variety of vendors to encourage cross platform connectivity.

Figure 5. Use case in which the client needed data, medical review, and operational oversight into their pivotal solid tumor oncology investigational products.
This combination of capabilities gives a company the opportunity to drill down in a clinical trial from the population level to one or a group of subjects. The resulting information reveals what needs to be reviewed or other required actions.
Many companies would benefit from these capabilities. In fact, the participants in a recent Applied Clinical Trial webcast sponsored by Revvity Signals noted that their most pressing challenges in querying clinical data include a lack of technical integration between electronic data capture and review, and the inability to efficiently track data review and query status. For electronic data capture in particular, this comes as no surprise, given that many systems lag behind the needs for automation and connectivity. Consequently, improvements in these tools could enhance their contribution to review processes.
CONCLUSION
The biopharma industry will continue to modify clinical trials, which will produce changes in the resulting data. With new kinds of data—likely more complex collections of data, and surely more data—biopharma companies will face increasing needs for software that overcomes challenges from data analysis to overall company performance and product quality. By centralizing analysis and communications, a biopharma company creates a system based on connectivity and automation, aggregated and usable data, flexible analysis, and methods of collaborative data review. This collection of capabilities is crucial in today’s biopharma industry and will be even more important in the future.

Meredith Nahm Zozus, PhD, CCDM
Professor and Director of Clinical Research Informatics
Joe R. and Teresa Lozano Long School of Medicine
University of Texas Health Science Center

Brent Meyers
Director, Clinical & Translational Analytics Product Portfolio
Brent Meyers has been with Signals Software since 2015 and currently leads the Clinical Solutions Product Portfolio. Brent has been building analytics solutions and leading technical delivery teams for over 18 years. Prior to his work at Signals Software, he has designed and built innovative clinical, operational, post-market, and other analytics solutions at IQVIA and PPD. Outside of the clinical trial space, Brent has also built analytic solutions for Northrop Grumman and led deployed technical teams in the U.S. navy. Brent holds a BA from The Citadel, an MBA from Meredith College, and currently resides in Portland, Oregon with his wife.
