Clinical Analytics
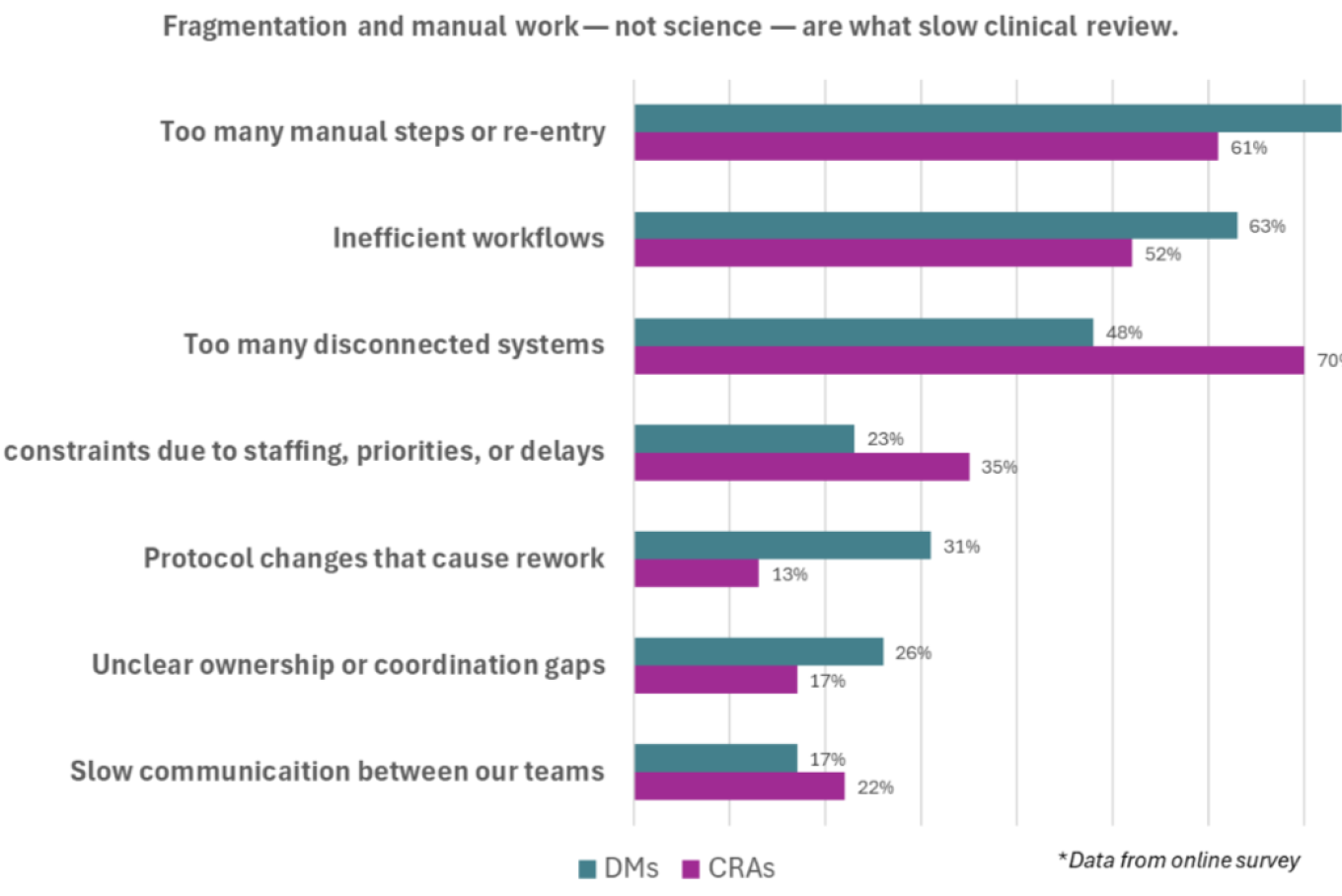
Reduce Review Delays Caused By Fragmented Clinical Data
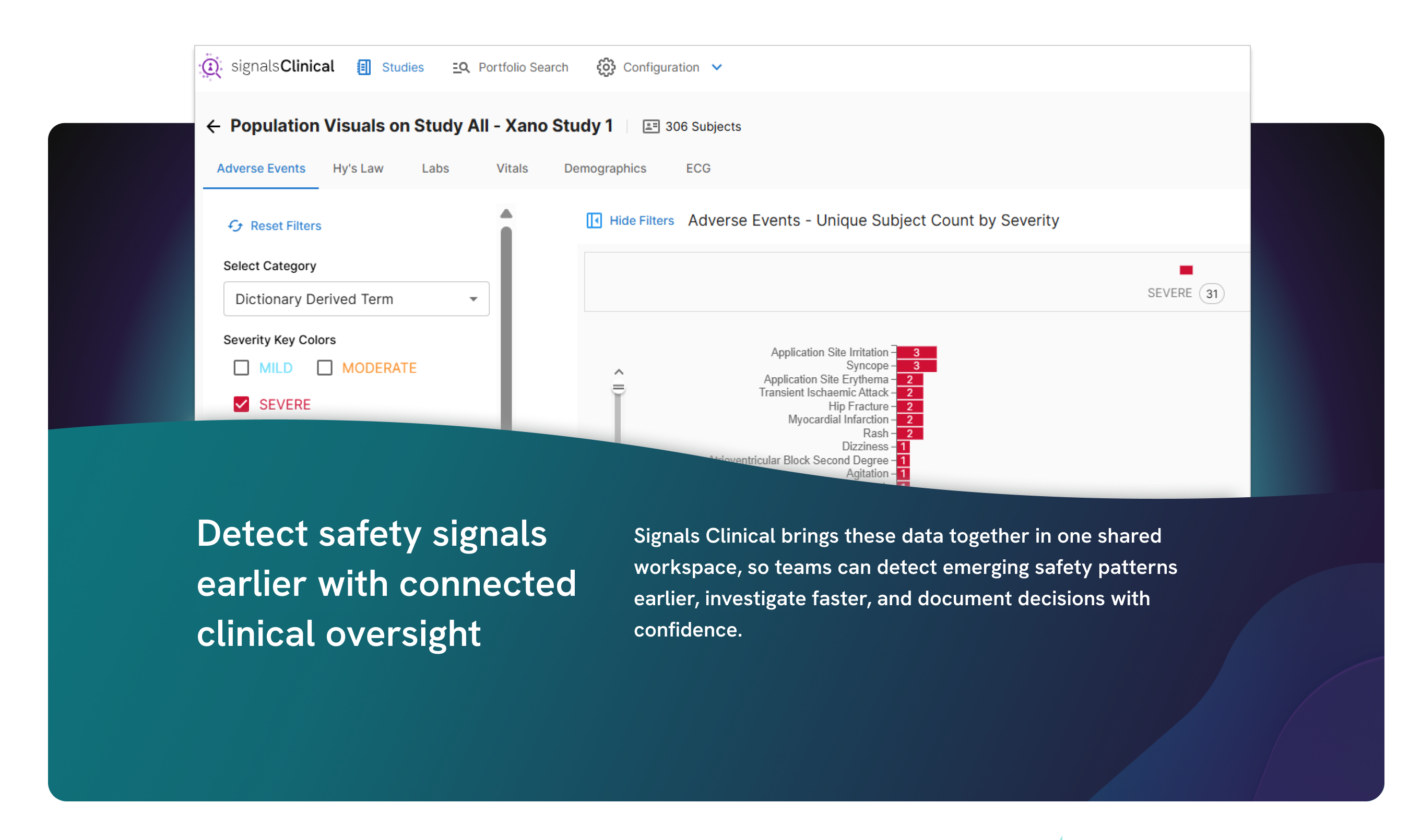
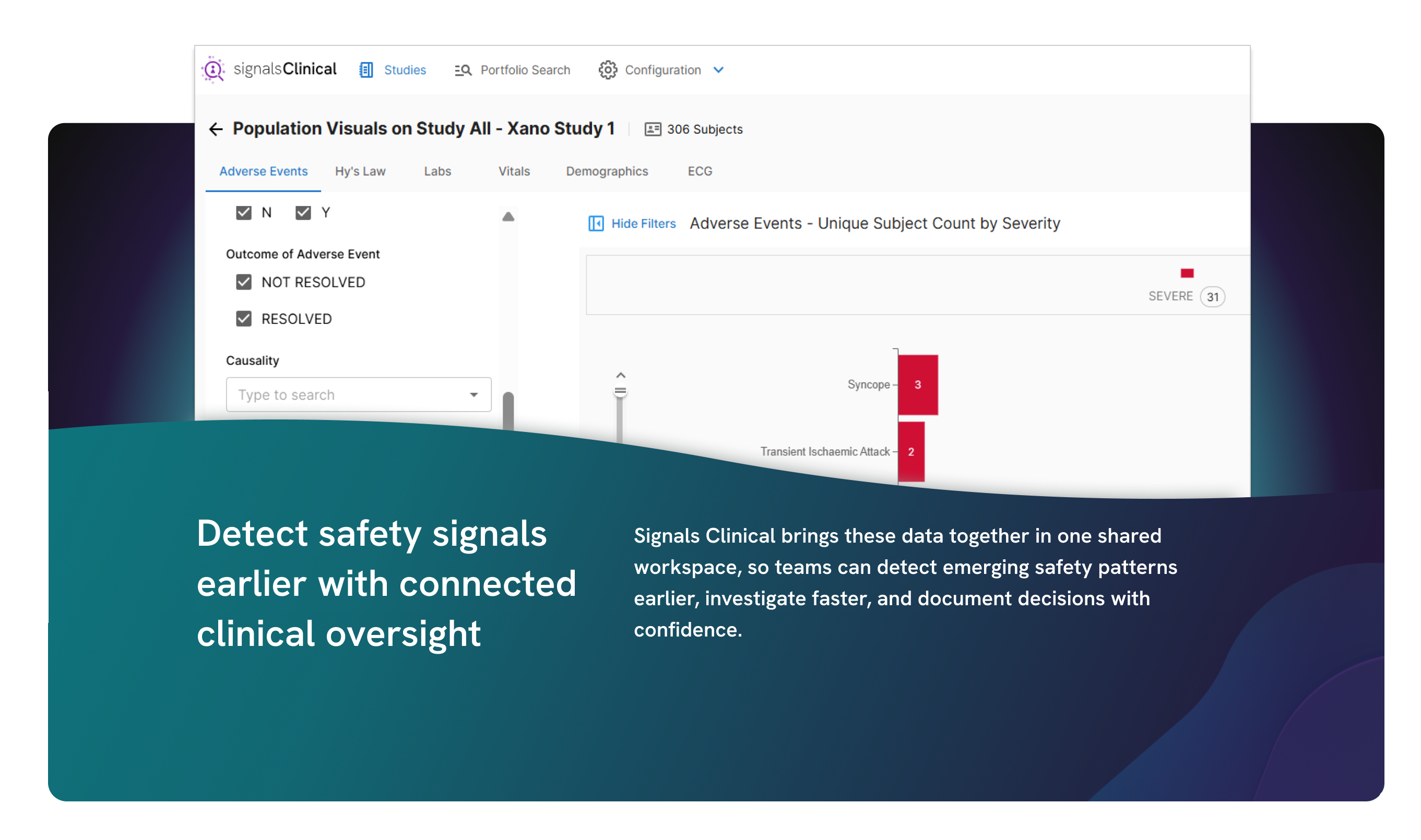
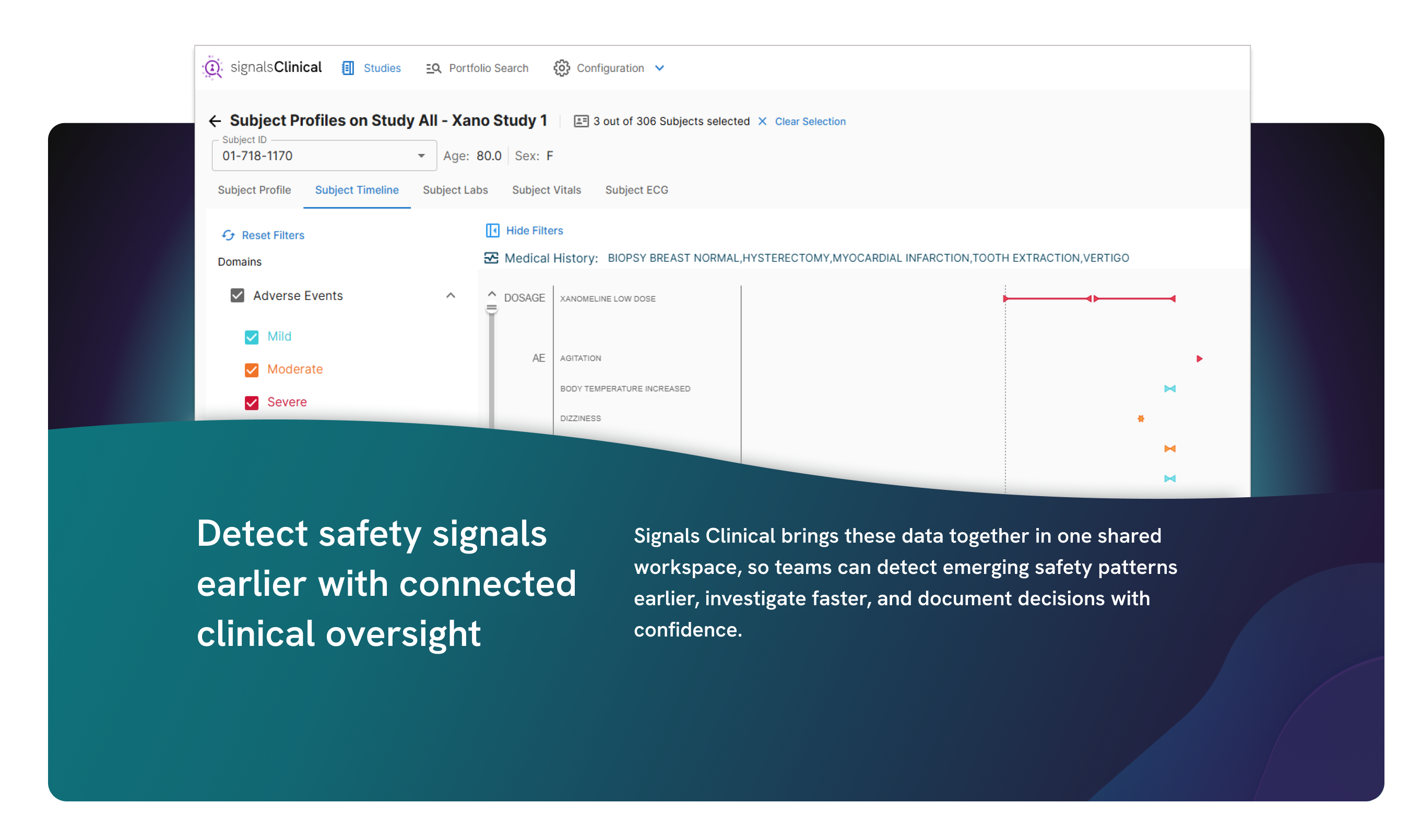
Bring all your clinical data into a single, unified view so you can streamline review, improve oversight, and make faster, defensible decisions across studies.
Trusted by Companies Worldwide
What Data Managers Can Achieve With Signals Clinical™
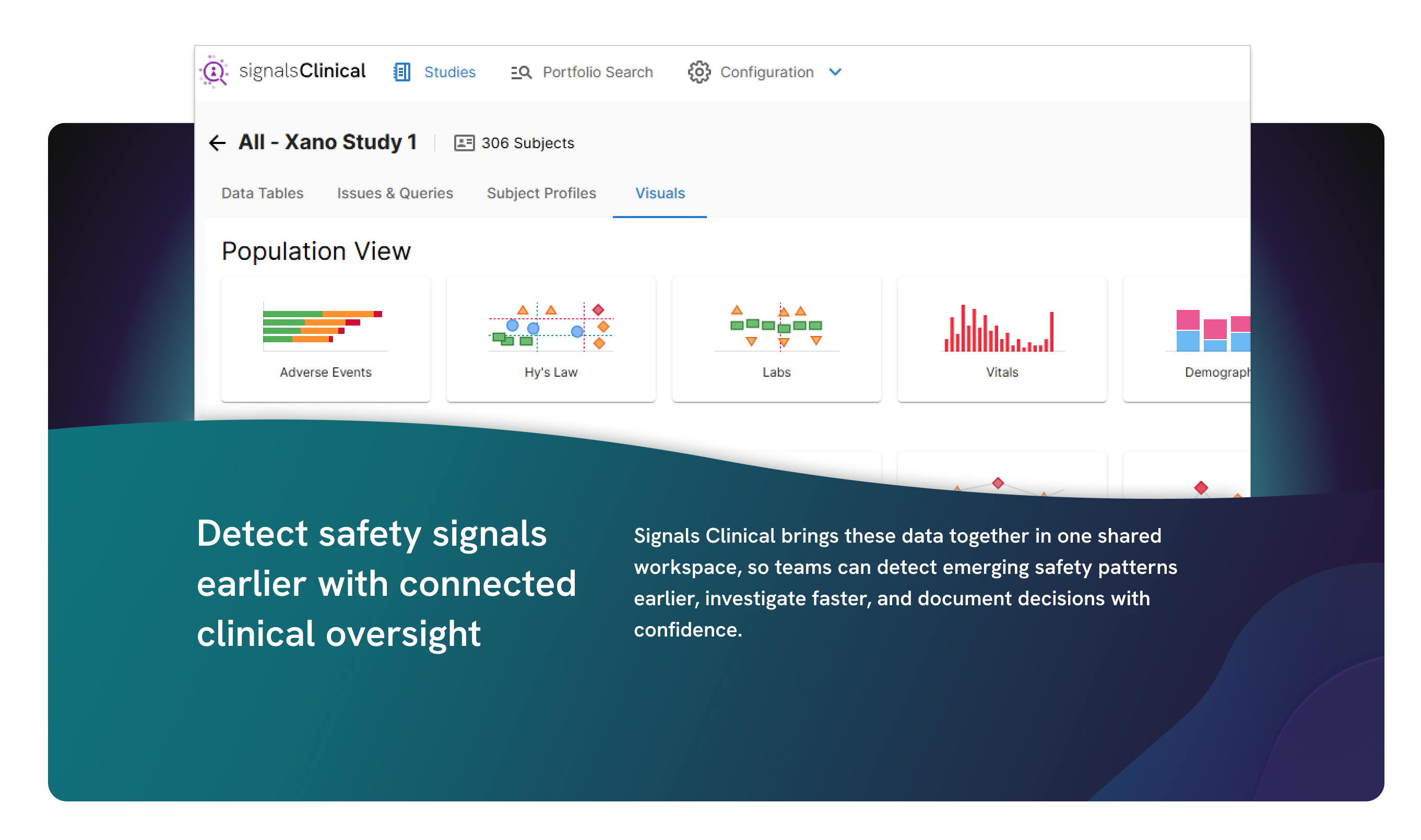
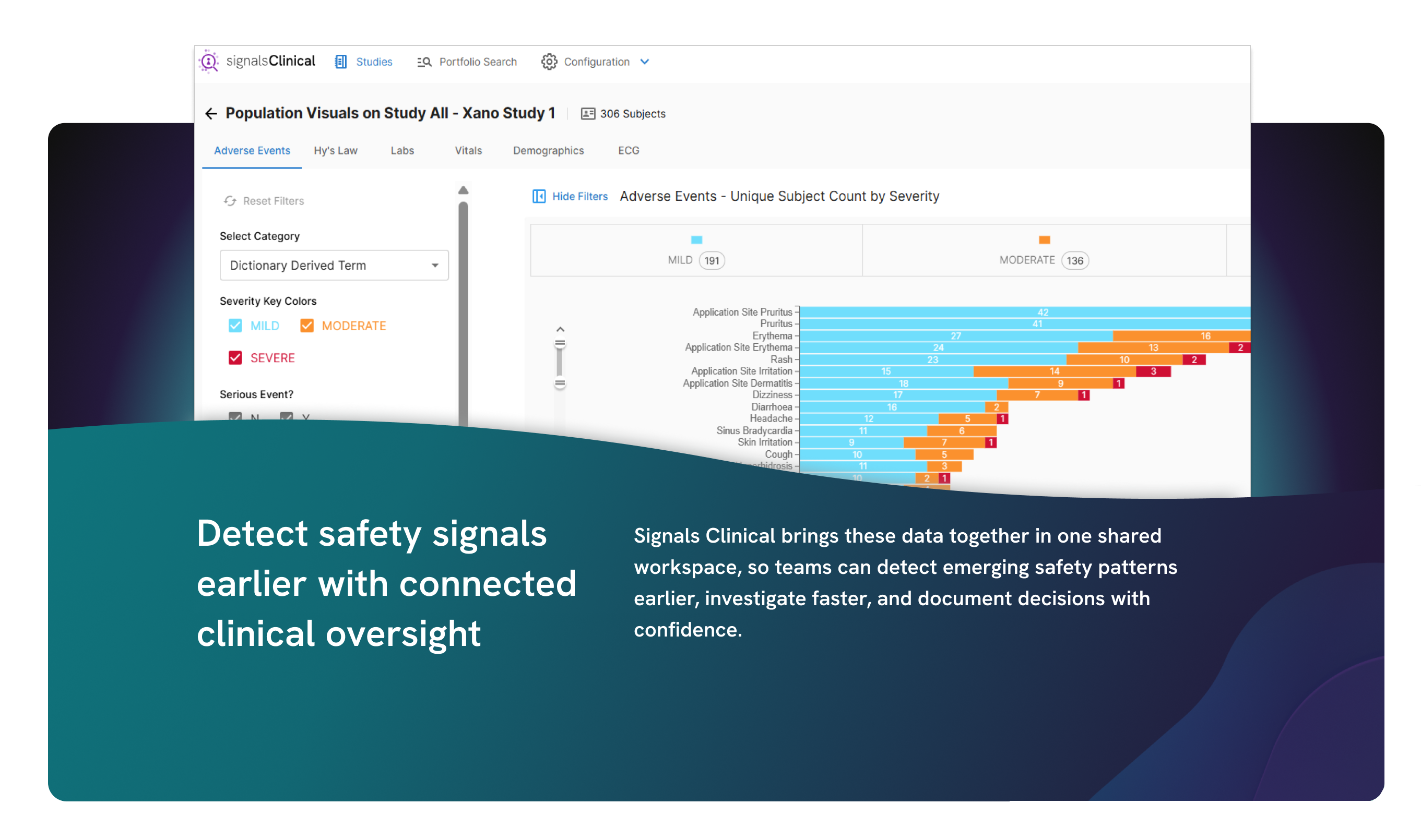
Adaptive analytics to accelerate clinical development
What Our Customers Are Saying
Resources
Keep up with the latest with guides, research, and insights.








Signals Clinical FAQs
Ready to take the next step?
Start a Guided Trial.
Experience Signals Clinical in a structured evaluation environment designed for real clinical review workflows. See how AI-assisted review accelerates listings, queries, and insights while maintaining traceability and oversight
Explore a Demo.
Watch how Signals Clinical transforms review—from rapid issue detection to real-time visual analytics powered by Spotfire®.
Talk to an Expert.
Connect with a specialist to explore your workflow needs and see how Signals Clinical can accelerate oversight and decision-making.