Overview
Since monoclonal antibodies (mAbs) were developed and received their first approval, they became indispensable as research toolsand biotherapeutic agents. In the course of their evolution, mAbs have been used in new ways, such as adding drug payloads to create antibody-drug conjugates (ADCs), merging two antibodies to make bispecific antibodies (bsAbs), and creating custom biomolecules from antibody fragments. AI technologies are enhancing and accelerating monoclonal antibody (mAb) and related therapeutic development, though their success depends on access to substantial volumes of< high-quality, relevant data.
This article will explore:
- The Pioneering First Approval
- Antibody Platforms Creating New Modalities
- Evolving the Clinical Development Lifecycle with AI and Data
Management - What’s Next for Antibody Development?
Introduction
The First Approvals
In the 1970s, immunologists invented a revolutionary technique to purify monoclonal antibodies (mAbs) to study how the adaptive immune system works.1 Sensing opportunity beyond research applications, clinical development yielded the first US Food and Drug Administration (FDA) approval for a therapeutic mAb in 1986.1 Muronomab was a CD3-directed mAb that targeted and depleted T cells and was initially approved to prevent organ transplant rejection.
It took 8 years for a second mAb to enter the therapeutic market, but the pace has increased exponentially since then. In the nearly four decades since the first approval, over 125 mAbs have been approved in various therapeutic areas. While oncology holds the lion’s share of approvals—41 of the first 100 approvals were for cancer indications1—mAbs are routinely used to treat blood clotting disorders, autoimmune diseases, allergic reactions, and more.1-4 Despite the skyrocketing pace of approvals and recent wins in neurodegeneration, therapeutic areas involving the brain and central nervous system, such as neurology, pain, and psychiatry, are still lacking in mAb treatment options.
The Antibody Platform Creating New Modalities
Advantageous Properties Drive Adoption
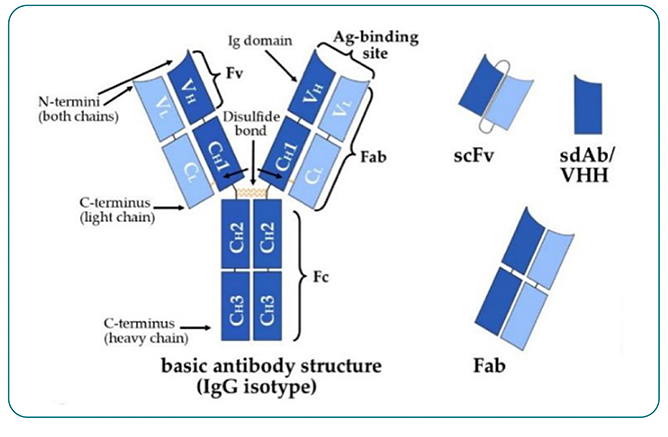
Who would have thought that antibodies — those Y-shaped proteins produced by B cells as the immune system’s molecular scouts — would have their natural role of recognizing foreign substances in the body harnessed to create one of the most powerful tools in modern drug development? Unmatched target specificity is the antibody’s superpower over other modalities: the development of mAbs was worthy of the 1984 Nobel Prize in Physiology of Medicine. As mAbs have become commonplace, some features are almost taken for granted. Their specificity allows mAbs to block signaling pathways, deplete target factors, and induce the immune system to kill cells through antibody-dependent cellular cytotoxicity (ADCC) and antibody-dependent cellular phagocytosis (ADCP). As effective as they are, whole mAbs are the crude first stage in targeted biotherapeutics. Evolving the class involves isolating their desirable properties, such as target-specificity and immune engagement, and tailoring them for new applications (Figure 1).
Antibody-drug conjugates (ADCs) and bispecific antibodies (bsAbs)
ADCs and bsAbs add new functionality to mAbs to expand their range of uses. ADCs are used in the clinic to deliver chemotherapeutic drugs that are not tissue specific. Enhertu® is an ADC built on the HER2-directed mAb trastuzumab that carries a cytotoxic topoisomerase inhibitor payload. The specificity of trastuzumab targets cancer cells over non-transformed cells, and the engineering of the linker and drug payload allows the inhibitor to dissociate from the mAb inside the target cells. This approach, like the development pattern of classical mAbs, started in cancer but is expanding into other indications, such as autoimmune disease, using drug payloads other than cytotoxic compounds.6
Unlike classical antibodies that have an affinity for one specific target, bsAbs are engineered to have two distinct antigen binding sites per molecule to bind two distinct targets. Bispecific constructs were conceived in the early days of mAb research and have taken many forms.7 The dual affinity allows bsAbs to be used in various ways, such as tethering target proteins or cells together or shuttling targets by secondary affinity to a transport protein (Figure 2). Emicizumab, approved in 2017, is a bsAb that binds clotting factors IX and X, serving as a functional replacement for clotting factor VIII that is missing in hemophilia A.8 Tethering immune cells to cancer-associated targets is the basis for a new wave of immuno-oncology therapeutics. CD3 is the most popular T cell-specific target and formed the basis of early T cell engagers, including blinatumomab—the first bsAb construct approved in oncology.1,9 The success of blinatumomab has accelerated interest in bsAbs, with over 300 ongoing and recruiting clinical trials (www.clinicaltrials. gov).10
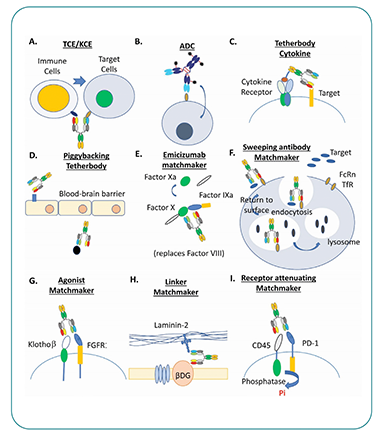
Figure 2. Examples of multispecific drug designs include T cell engagers/ NK cell engagers (TCE/KCE), ADCs, and various “matchmaker” bispecifics, including emicizumab. Image source: Zhong and D’Antona (2021)11
The uses of bsAbs include tethering targets together, improving specificity by requiring simultaneous binding of multiple proteins, and a range of “matchmaking” applications (Figure 2).11 Further bsAb innovations include trispecific antibodies created by fusing additional antibody fragments onto a bsAb construct. The potential uses for multivalent antibodies include extending serum half-life, activating co-stimulatory pathways for more efficient immune cell engagement, and providing a logic gate to enhance target specificity and reduce the chances of immune escape by the target cells.12,13 Another innovative use is using one of the two bispecific paratopes to target transport proteins to improve access to targets, such as using transferrin to shuttle the antibody across the blood- brain barrier to reach targets inaccessible to current intravenous biologics.1,5
Antibody Fragments: Fabs and ScFvs
Antibody fragments are derivatives designed to retain antigen binding in a smaller form than the parent antibody and include antigen-binding fragments (Fab) and single-chain variable fragments (ScFvs) (Figure 3). The first approved antibody fragment was abciximab, a Fab targeting αIIb 3 integrin to prevent blood clots, and three more have been approved since 1994.14 ScFvs are smaller fragments than Fabs, and include brolucizumab, a standalone ScFv approved in macular degeneration.
ScFvs have been used extensively in research or as a building block in larger peptide constructs. Most of the seven approved ScFvs are incorporated into larger engineered protein structures.14 Blinatumomab is a bispecific T cell engager (BiTE) composed of two linked ScFvs, one targeting CD3 and the other targeting CD19, to treat relapsed and/or refractory B cell precursor acute lymphoblastic leukemia (B-ALL).9 In addition, ScFvs are being used as part of engineered chimeric antigen receptors (CARs), which grant CAR-T cell therapies their target specificity.15
ScFvs are also used in novel immune-engaging technologies: targeting major histocompatibility complex bound with a target peptide (pMHC) would enable antibodies and antibody fragments
to recognize peptides that are typically inside cells.16 One approach is to develop antibodies that recognize the target pMHC, referred to as T cell receptor (TCR) mimic antibodies.17 Another technology to target pMHC uses a CD3-directed ScFv linked to an engineered recombinant TCR to create T cell engagers called ImmTACs (Immune mobilizing monoclonal T-cell receptors Against Cancer).9,18,1
Single-domain Antibodies: Nanobodies
Nanobodies are derived from heavy chain-only camelid antibodies and are the smallest naturally derived antigen-binding fragment (See Figure 3).9,20 In 2018, the FDA approved the first nanobody therapeutic: caplacizumab to treat adults with acquired thrombotic thrombocytopenic purpura.9
Like with ScFvs, nanobodies are often used as building blocks for multivalent constructs: BI836880 is a tri specific construct targeting VEGF and angiopoietin-2 to block ligand-receptor interactions and human serum albumin to extend half-life.21While antibodies, Fabs, and ScFvs have been used mainly to block interactions, nanobodies, with their small size, have been explored as protein-based receptor agonists and allosteric modulators, allowing isoform and dimerization-status specificity for targeting G-protein coupled receptors.22,23
Evolving the Clinical Development Lifecycle with AI and Data Management
Developing a therapeutic antibody product is complex and expensive, requiring a go-to-market strategy that de-risks development. Historically, mAbs were developed for diseases of high unmet need and then expanded into other therapeutic areas. An example is Rituximab, a CD20-targeting mAb originally approved to treat B cell lymphomas. As the role of B cells in autoimmune diseases became evident, rituximab earned approvals across autoimmune and rheumatoid diseases and continues to be explored in additional indications.24–26 Rituximab also shows how a strong biological rationale remains the guiding light for antibody development, especially as AI and data management systems augment and streamline development.
AI-Augmented Antibody Development
The first fully AI-developed antibody, bimekizumab, is already on the market and shows how AI techniques can augment hypothesis-driven research.27 Researchers hypothesized that blocking both IL-17A and IL-17F would be an effective strategy to target plaque psoriasis, more so than targeting either alone. However, the hunt for antibody binders of both IL-17A and IL-17F using traditional approaches and screening billions of candidates came up empty. Using an AI model trained with data from the screened candidates, 18 new candidates were proposed, and one became bimekizumab without further modification.27 AlphaFold, Developers won the Chemistry Nobel Prize in 2024, the premiere generative AI protein language model trained on the Protein Database (PDB.org) structures, has become a powerful tool for predicting protein structures and protein- protein interactions.28,29 Beyond the AI-powered design of biomolecules like bimekizumab, one of the impactful uses of AI in biologics discovery is narrowing the search for candidates. Rather than screening possibly billions of antibody candidates, tools like AlphaFold can streamline development by identifying promising candidates in silico to narrow the candidate pool, saving time and resources.27–30 The excitement around AI and its potential in biotherapeutics development is producing intense investment. Biopharma giants, including AbbVie, AstraZeneca, and Sanofi, partnered with AI-driven protein design companies in 2023 to integrate AI capabilities into their wet lab expertise.31 The AI race shows no signs of slowing down, as groups in academia and industry develop models that not only predict structures from protein sequences, but also predict protein sequences likely to produce desired structures and characteristics.32,33 Evolutionary Scale showed their proof of principle by designing entirely new fluorescent proteins based on the chromophore- containing barrel structure of GFP, but with vastly different sequences.33 With these advances, generative AI has the potential to produce antibodies against never-before-seen targets, given the vast protein sequence and structure databases available for training.30
What’s Next for Antibody Development?
Taming the Immune Response
ADCC was a feature exploited in oncology in developing biologics, like trastuzumab, that direct the immune system to neutralize the “tagged” cells. We are learning how the Fab domain affects immune-related cytotoxicity and how we can tune the potency of the Fc domain.34 Increased ADCC and ADCP are desirable outcomes for cancer treatment, but for indications where the antibody is used as a signal transduction blocker, such as in autoimmunity, cytotoxicity is not needed. Immune cell engagement has become a hot field in oncology with the increasing development of T cell engagers, CAR-T therapies, and TCR-based therapeutics. CAR-T therapies release cytokines to recruit the immune system to join the fight, while BiTEs and bispecifics help activate T cells by showing them their targets.
Broadening Antibodies as Delivery Vehicles
ADCs, typically associated with delivering cytotoxic payloads, are being adapted for non-oncological applications.35 One application being explored is using ADCs to deliver drugs across the blood-brain barrier by tying the drug payload to an antibody targeting transferrin for transport.5 Another application is delivering steroids to sites of action rather than broad systemic administration, which is problematic for long-term use in autoimmune and rheumatic disease.
The AI Race will Bring New Modalities
With a successful AI-generated biotherapeutic agent on the market, leaders in the biotech industry envision a future with “modality-agnostic” AI-supported discovery.27 We’ve already seen that AI can identify patterns in protein interactions and suggest modifications that researchers would not have considered, such as altering sequences outside the antibody paratope.36 AI-driven protein folding and docking predictions can already aid in directing biomolecule design, focusing research on promising candidates, and modifying existing ones.23,30,37 EvolutionaryScale’s models also demonstrate that researchers may soon be able to tell a computer what kind of protein or antibody they want to make and quickly get a prototype to start optimizing.32,33

However, the quality of AI- driven insights hinges on having AI-ready data from high-quality basic research. The insights leading to bimekizumab would not have happened if not for the millions of characterized candidates used to train the AI model to augment discovery.
AI may also unlock the potential of modalities that have not yet lived up to their promise. Fab and ScFv antibody fragments have been hypothesized to penetrate tissue more effectively than their parent mAbs, but they have not yet shown the anticipated benefits over full-sized mAbs. AI- driven protein modeling could help design effective fragments and even smaller, target- specific biomolecules and drug delivery methods. Affibodies, derived from the immunoglobulin- binding Z domain of bacterial protein A, are small (~6 kDa versus 25 kDa for ScFv and 50 kDa for Fab fragments) and have already seen applications in radiolabeling of PD-L1 expression.38 AI-powered protein design could see antibody and antibody- adjacent modalities like this explode with potential applications.
Revvity Signals Perspective
Antibody drug discovery is inherently complex, especially with the staggering assortment of new
antibody formats in preclinical development and entering clinical testing.5 Antibody development requires seamless coordination between teams, technologies, and data at every stage— from designing and testing to improving candidates. Scientists face challenges identifying liabilities, interpreting vast datasets, and iterating quickly to make confident decisions. At the same time, R&D leaders need a comprehensive, real-time view of antibody candidates to holistically evaluate assets, select criteria, and prioritize the most promising paths forward.

Revvity Signals addresses these challenges head-on, providing scientists and leaders with the tools they need to accelerate the antibody discovery process. The Antibody Developability Dashboard, powered by Spotfire, places bioinformatics at scientists’ fingertips, enabling rapid analysis of antibody sequences for potential liabilities or optimization needs.
By reducing turnaround time for “design-make-test-decide” cycles, scientists can fail fast helping to identify areas for improvement, ensuring assets progress through the pipeline efficiently.
Leaders, meanwhile, can bring together scientific disciplines across modalities ex: antibodies and small molecules. This end to- end research “under one roof” allows them access to holistic insights through intuitive dashboards and tools within Signals One, allowing them to compare candidates, apply expert criteria, and focus on assays or assets that matter most. Whether narrowing down options, deciding to move forward, or re-prioritizing experiments, the ability to visualize data and insights in a unified way transforms decision-making processes.
With Signals One, Revvity Signals bridges the gap between complex bioinformatics, collaboration, and actionable insights — empowering teams to overcome discovery hurdles and drive antibody development with greater speed, clarity, and confidence.
References
1. Mullard A. FDA approves 100th monoclonal antibody product. Nature Reviews Drug Discovery. 2021;20(7):491-495. doi:10.1038/d41573-021-00079-7
2. Strohl WR. Structure and function of therapeutic antibodies approved by the US FDA in 2023. Antibody Therapeutics. 2024;7(2):132-156. doi:10.1093/abt/tbae007
3. Cohen F. Alzheimer’s: FDA approves Eli Lilly drug donanemab for amyloid plaques. Medical News Today. April 3, 2024. Accessed July 5, 2024. https://www.medicalnewstoday.com/articles/fda-approves-alzheimers-drug-donanemab
4. Delrieu J, Andrieu S, Vellas B. Dementia research in 2023: the year of anti-amyloid immunotherapy. The Lancet Neurology. 2024;23(1):13-15. doi:10.1016/S1474-4422(23)00449-0
5. Elshiaty M, Schindler H, Christopoulos P. Principles and current clinical landscape of multispecific antibodies against cancer. International Journal of Molecular Sciences. 2021;22(11):5632. doi:10.3390/ijms22115632
6. McPherson MJ, Hobson AD, Hernandez A, et al. An anti–TNF–glucocorticoid receptor modulator antibody-drug conjugate is efficacious against immune-mediated inflammatory diseases. Science Translational Medicine. 2024;16(739):eadd8936. doi:10.1126/scitranslmed.add8936
7. Labrijn AF, Janmaat ML, Reichert JM, Parren PWHI. Bispecific antibodies: a mechanistic review of the pipeline. Nat Rev Drug Discov. 2019;18(8):585-608. doi:10.1038/s41573-019-0028-1
8. Prezotti ANL, Frade-Guanaes JO, Yamaguti-Hayakawa GG, Ozelo MC. Immunogenicity of current and new therapies for hemophilia A. Pharmaceuticals (Basel). 2022;15(8):911. doi:10.3390/ph15080911
9. Jin S, Sun Y, Liang X, et al. Emerging new therapeutic antibody derivatives for cancer treatment. Sig Transduct Target Ther. 2022;7(1):1-28. doi:10.1038/s41392-021-00868-x
10. Center for Drug Evaluation and Research. Bispecific antibodies: an area of research and clinical applications. FDA. February 14, 2024. Accessed June 17, 2024. https://www.fda.gov/drugs/spotlight-cder-science/bispecific-antibodies-area-research-and-clinical-applications
11. Zhong X, D’Antona AM. Recent advances in the molecular design and applications of multispecific biotherapeutics. Antibodies. 2021;10(2):13. doi:10.3390/antib10020013
12. Klein C, Brinkmann U, Reichert JM, Kontermann RE. The present and future of bispecific antibodies for cancer therapy. Nat Rev Drug Discov. 2024;23(4):301-319. doi:10.1038/s41573-024-00896-6
13. Meetze K, Mehta NK, Li B, et al. CLN-978, a novel half-life extended CD19/CD3/HSA-specific T cell-engaging antibody construct with potent activity against B-cell malignancies with low CD19 expression. J Immunother Cancer. 2023;11(8):e007398. doi:10.1136/jitc-2023-007398
14. Antibody therapeutics approved or in regulatory review in the EU or US. The Antibody Society. 2024. Accessed July 6, 2024. https://www.antibodysociety.org/resources/approved-antibodies/
15. Alnefaie A, Albogami S, Asiri Y, et al. Chimeric antigen receptor T-cells: an overview of concepts, applications, limitations, and proposed solutions. Frontiers in Bioengineering and Biotechnology. 2022;10. https://www.frontiersin.org/articles/10.3389/fbioe.2022.797440
16. Wieczorek M, Abualrous ET, Sticht J, et al. Major histocompatibility complex (MHC) class I and MHC class II proteins: conformational plasticity in antigen presentation. Front Immunol. 2017;8:292. doi:10.3389/fimmu.2017.00292
17. Duan Z, Ho M. T cell receptor mimic antibodies for cancer immunotherapy. Mol Cancer Ther. 2021;20(9):1533-1541. doi:10.1158/1535-7163.MCT-21-0115
18. Immunocore. Science. Immunocore Holdings plc. 2024. https://www.immunocore.com/science
19. Immunocore reports updated Phase 1 data of brenetafusp (IMC-F106C), an ImmTAC bispecific targeting PRAME, in immune checkpoint pre-treated cutaneous melanoma patients at ASCO 2024. Immunocore Holdings plc. May 31, 2024.
20. Kijanka M, Dorresteijn B, Oliveira S, van Bergen en Henegouwen PMP. Nanobody-based cancer therapy of solid tumors. Nanomedicine (Lond). 2015;10(1):161-174. doi:10.2217/nnm.14.178
21. Hofmann I, Baum A, Hofmann MH, et al. Pharmacodynamic and antitumor activity of BI 836880, a dual vascular endothelial growth factor and angiopoietin 2 inhibitor. J Pharmacol Exp Ther. 2023;384(3):331-342. doi:10.1124/jpet.122.001255
22. Cromie KD, Van Heeke G, Boutton C. Nanobodies and their use in GPCR drug discovery. Curr Top Med Chem. 2015;15(24):2543-2557. doi:10.2174/1568026615666150701113549
23. Haubrich J, Font J, Quast RB, et al. A nanobody activating metabotropic glutamate receptor 4 discriminates between homo- and heterodimers. Proc Natl Acad Sci USA. 2021;118(33):e2105848118. doi:10.1073/pnas.2105848118
24. Garcia-Montoya L, Villota-Eraso C, Yusof MYM, Vital EM, Emery P. Lessons for rituximab therapy in patients with rheumatoid arthritis. The Lancet Rheumatology. 2020;2(8):e497-e509. doi:10.1016/S2665-9913(20)30033-3
25. Pierpont TM, Limper CB, Richards KL. Past, present, and future of rituximab—the world’s first oncology monoclonal antibody therapy. Front Oncol. 2018;8:163. doi:10.3389/fonc.2018.00163
26. Chen YH, Wang XY, Jin X, Yang Z, Xu J. Rituximab therapy for primary Sjögren’s syndrome. Front Pharmacol. 2021;12:731122. doi:10.3389/fphar.2021.731122
27. Zhavoronkov A. AI in big pharma: the first antibody designed using AI in the clinic. Forbes. December 21, 2022.
28. Abramson J, Adler J, Dunger J, et al. Accurate structure prediction of biomolecular interactions with AlphaFold 3. Nature. Published online May 8, 2024. doi:10.1038/s41586-024-07487-w
29. AlphaFold. Google DeepMind. October 13, 2022. https://deepmind.google/technologies/alphafold/
30. Shanehsazzadeh A, McPartlon M, Kasun G, et al. Unlocking de novo antibody design with generative artificial intelligence. 2024. doi:10.1101/2023.01.08.523187
31. Biopharma Dealmakers. Antibody design enters the AI era. Published online May 31, 2024. doi:10.1038/d43747-024-00030-w
32. Shanker VR, Bruun TUJ, Hie BL, Kim PS. Unsupervised evolution of protein and antibody complexes with a structure-informed language model. Science. 2024;385(6704):46-53. doi:10.1126/science.adk8946
33. Callaway E. Ex-Meta scientists debut gigantic AI protein design model. Nature. Published online July 8, 2024. doi:10.1038/d41586-024-02214-x
34. Kang TH, Jung ST. Boosting therapeutic potency of antibodies by taming Fc domain functions. Exp Mol Med. 2019;51(11):1-9. doi:10.1038/s12276-019-0345-9
35. Pal LB, Bule P, Khan W, Chella N. An overview of the development and preclinical evaluation of antibody–drug conjugates for non-oncological applications. Pharmaceutics. 2023;15(7):1807. doi:10.3390/pharmaceutics15071807
36. Callaway E. How generative AI is building better antibodies. Nature. 2023;617(7960):235. doi:10.1038/d41586-023-01516-w
37. Eshak F, Pion L, Scholler P, et al. Epitope identification of an mGlu5 receptor nanobody using physics-based molecular modeling and deep learning techniques. J Chem Inf Model. 2024;64(11):4436-4461. doi:10.1021/acs.jcim.3c01620
38. Grindel BJ, Engel BJ, Ong JN, et al. Directed evolution of PD-L1-targeted affibodies by mRNA display. ACS Chem Biol. 2022;17(6):1543-1555. doi:10.1021/acschembio.2c00218

End-to-end workflow support across scientific disciplines.

The Standard for Chemical Drawing.

End-to-end Clinical Data Science Platform.
