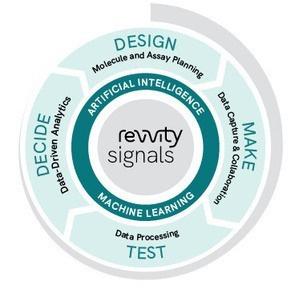
A unified, cloud-native SaaS solution that drives scientific collaboration across Drug Discovery
Complexity in New Drug Modalities Requires Multidisciplinary Collaboration
Emerging classes of therapeutics exacerbate the challenges in drug discovery because they often force the rapid adoption of new assay techniques or require different data representations. Partnerships between biologists and medicinal chemists are driving development in novel drug modalities, including technological advances in protein degraders, peptide therapeutics, antibody development, cell and gene therapies, and RNA-based therapeutics.
Supporting these new scientific endeavors requires efficient collaboration across multidisciplinary, globally distributed teams. These dispersed teams can collaborate efficiently with cloud-based, workflow-focused solutions.
Challenges and Opportunities in Integrating Informatics for Drug Discovery
Historically, drug discovery information has been captured in self-managed informatics systems, using bespoke software targeting individual subdomains of the overall process. These include medicinal chemistry, lead optimization, in vitro assays, cell line optimization, biologics process optimization, in vitro drug metabolism and pharmacokinetics (DMPK ) assays, animal pharmacokinetic studies, and animal efficacy and safety studies. The fragmented nature of this informatics environment has created several key challenges:
- Data integration. New biological and chemical entity (NBE or NCE) discovery generates vast amounts of data from various sources, including experiments, literature, and databases. Integrating and analyzing this data is a challenging task that requires new data management and analysis tools.
- Data quality. Quality data are necessary for making accurate and reliable predictions. Intra-source variability can make it challenging to integrate and analyze data effectively.
- Machine learning. Machine learning (ML) has become an essential tool in drug discovery, aiding researchers in predicting the properties of new molecules and identifying targets. Developing accurate and reliable ML models requires high-quality data and sophisticated algorithms.
- Visualization. NBE and NCE discovery generates complex data that can be difficult to interpret and analyze. Effective data visualization tools enable researchers to identify patterns and trends in the data and make more informed decisions.
- Legal considerations. The use of informatics in drug discovery raises several legal concerns, including data privacy, intellectual property, and regulatory compliance. Researchers must conduct informatics work in compliance with relevant laws and regulations.
- Collaboration. Effective collaboration tools and platforms are necessary to facilitate research and accelerate drug discovery.
Advanced Workflow Focused Solutions are Critical
Introducing Signals One™ for Drug Discovery
One solution for Design-Make-Test-Decide
Design & Make
Signals One is intuitive. It has DNA fluency for biologists, HELM integrations to support the bridge between chemically modified biopolymers and is natively , integrated with ChemDraw to allow chemists to properly document their work. The notebook digitally maintains scientific integrity and encourages creativity and productive thinking, all while fostering collaboration across multidisciplinary teams. Finally, it fundamentally supports intellectual property protection for patents.
- Data are easily accessible from one location
- Automated stoichiometric calculations
- Search for prior knowledge
- Material inventory management and safety information
- Access controls enable group-specific data access
- Support for patent applications
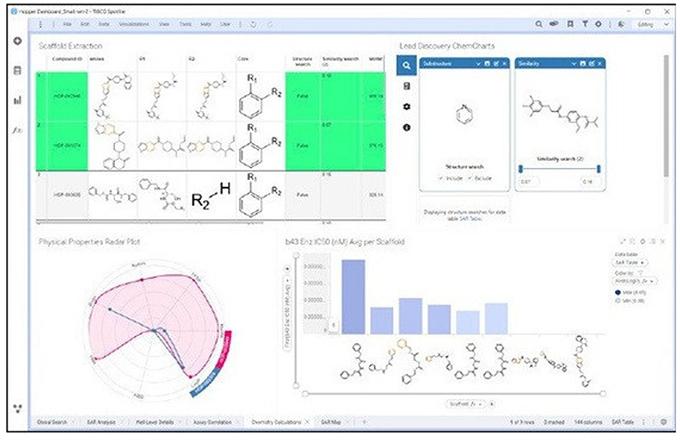
Figure 2. The first step the Design-Make-Test-Decide R&D workflow. Signals One can be used to plan, capture data, and collaborate.
ChemDraw®, the trusted chemistry gold standard for the last 35+ years, has been continuously upgraded to include colorful, interactive features to make high quality chemical drawings and 3D models, including the ability to copy 3D figures into PowerPoint® (3MF copy) to enhance communication with collaborators and stakeholders. ChemDraw is the chemical brain that natively powers the Signals One.
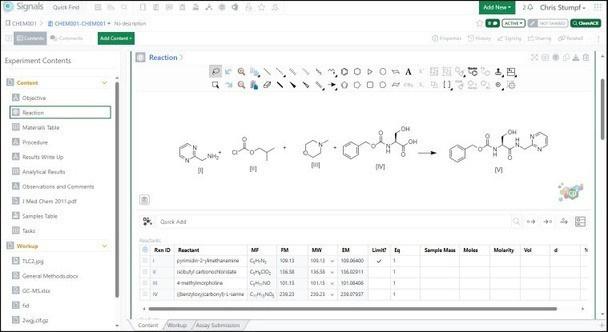
Figure 3. Native ChemDraw integration in Signals One. Chemical synthesis reactions can be easily drawn using ChemDraw and stoichiometry tables automatically update.
Hierarchical Editing Language for Macromolecules (HELM), another new Signals One feature, facilitates accurate visual representation of new drug modalities by allowing investigators to include amino acids, peptides, and nucleic acids in core molecular structures. Researchers can communicate and manage biomolecules through shared libraries in Signals One. This enables biologists and chemists to accurately, and easily represent modified small- to medium-sized biomolecules and all their associated scientific testing in one easy-to-use place.
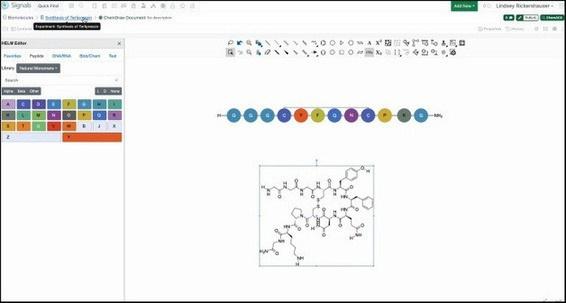
Figure 4. Using the HELM Editor in ChemDraw to draw Terlipressin in amino acid notation (top) and conversation of the amino acids into a chemical structure (bottom).
Test
Signals One includes two-levels of data processing capabilities to provide a flexible environment that caters to a wide range of analytics needs: For routine use cases including in vitro curve fitting, in vivo data analysis, or group comparisons, Signals One keeps simple analytics simple with native analytics that is designed to be intuitive.
For advanced use cases, where research applications are more complex, researchers can use the data processing powered by Spotfire to analyze data and convert complex results into intuitive graphics. This facilitates unified assay data management, rapid assessment of hit compounds, and graphing of IC curves, kinetics, and other results from critical assays to evaluate efficacy and determine whether modifications are needed. The data processing capability facilitates templatizations for faster analysis and the standardization of analysis practices across researchers and research teams contributing to data consistency.
- One software application for unified assay data management covering multiple instruments and assay types saving time and increasing data accuracy.
- Workflow templates are set up once and used multiple times.
- Graphics driven by Spotfire™ enable meaningful visualization of data.
- Standardized Statistical Analysis provides cluster analysis and unsupervised ML.
- Manage vast amounts of data.
- Simplified reporting pushes directly into Signals One and exports into Microsoft Office and PDF format.
- Automatic round trip of experimental results from the data processing capability into Signals One
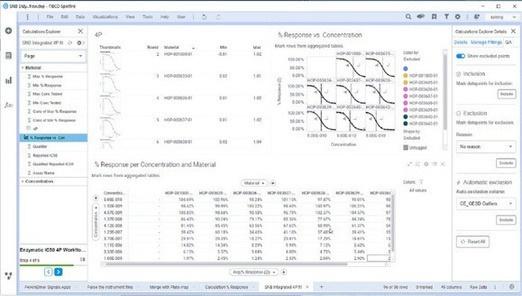
Figure 5. The Data processing capability represents the Test phase of the Design-Make-Test-Decide R&D workflow. The capability provides guided workflows for data import and analysis so that visualization and analytics are consistent and reliable.
Decide
The data-driven analytics capability provides next-generation data management, guided search, and advanced Structure Activity Relationship (SAR) analysis. It features an intuitive point-and-click query builder to access, parse, and analyze large datasets without the need for writing code. Biologists and chemists can visualize every experiment and project holistically, gaining unique, unforeseen insights critical to the discovery process in hours instead of months. This facilitates the advancement of molecules to clinical trials, ultimately increasing the chances of bringing a new drug to market.
The Only SAR Software Application with Native ChemDraw Integration
One Team, One Goal – Harmonized Alignment for Scientific Excellence
Better Data, Insights, and Outcomes

Signals One is the first fully integrated SaaS solution for new biological and chemical modalities research, ideally suited for research use cases involving early and late-stage in vitro assays, cell line optimization, biologics process optimization, in vitro DMPK assays, animal pharmacokinetic studies, stage animal efficacy and safety studies. Signals One is an ideal solution for scientists working on new drug modalities or who want to expand the power of Signals Notebook across the entire Design-Make-Test-Decide R&D lifecycle for better data, insights, and outcomes.
To learn more about the Signals One by Revvity Signals for new biological and chemical modalities research and to increase your chance for market success, visit: https://revvitysignals.com/products/research/signals-one

End-to-end workflow support across scientific disciplines.

The Standard for Chemical Drawing.

End-to-end Clinical Data Science Platform.