Signals Clinical™
Use AI Augmented Clinical Data Review To Gain Oversight You Can Stand Behind
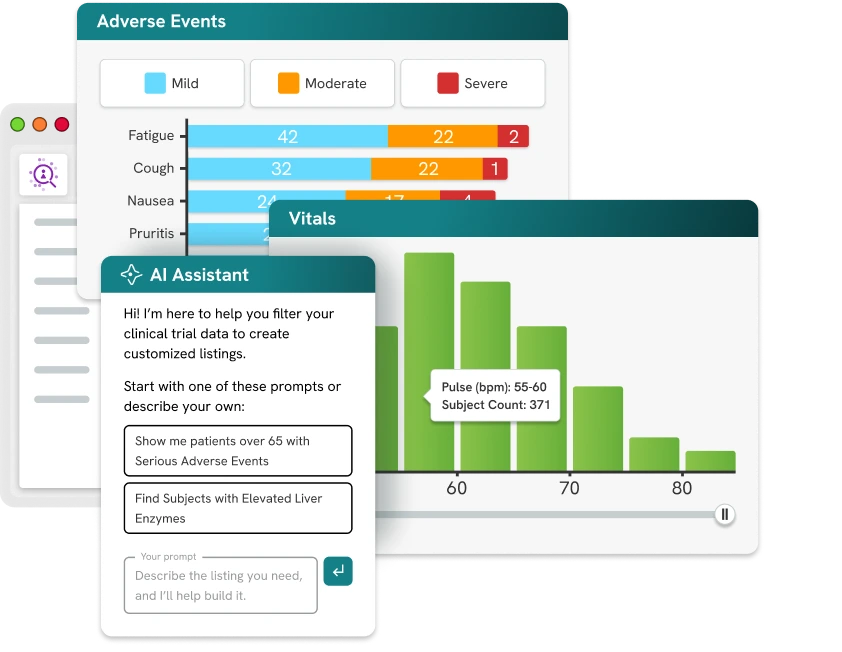
Bring your listings, queries, and medical monitoring into one AI‑assisted workspace so you can analyze studies flexibly, streamline review, and maintain compliance‑ready oversight all with Spotfire® built in.
Trusted by Companies Worldwide
Accelerate Clinical Decisions with AI Assisted Insight
Adapt your trials with prebuilt visualizations and flexible, study‑specific analytics powered by Spotfire® for any protocol, portfolio, or partnership model.
Prebuilt clinical visualizations
Detect anomalies faster with ready‑to‑use dashboards for labs, vitals, AEs, and more.
AI-assisted queries and issues
Track, prioritize, and resolve issues with complete visibility across sites and studies.
Traceable, compliance-ready workflows
Log every review action to support sponsor oversight and inspection‑ready traceability.
Real-time clinical insights
Move beyond static listings with live data views that keep pace with your trials.
Built for your clinical teams
Clinical Data Manager
Reduce manual prep, standardize listings, and launch reviews faster. Use prebuilt visuals and AI-assisted workflows to surface issues early and streamline query cycles.
Medical Monitor / Safety Physician
Get a clearer view of subject safety across labs, vitals, and AEs with interactive visualizations that highlight patterns, outliers, and emerging risk.
Clinical Operations Lead / Sponsor
Monitor study performance and oversight activities across portfolios, CROs, and regions in one place with standardized, traceable review workflows.
Solutions for clinical research and development teams
Emerging Biotechs
Start fast with prebuilt clinical visualizations and templates designed for first-in-human and early-phase trials, without needing a large data infrastructure.
Growing Biopharma
Scale clinical analytics across multiple studies and indications with reusable workflows, portfolio dashboards, and integrated Spotfire® analytics.
CROs & Enterprises
Enable shared oversight, standardized review processes, and sponsor-facing dashboards across dozens or hundreds of trials.
Security
Secure Data
Data encrypted in transit and at rest to protect sensitive clinical information.
Role-Based Access
Fine-grained permissions ensure the right stakeholders see the right studies and datasets.
Audit-Ready Logs
Automatic tracking of actions and changes supports inspections and internal QA reviews.
Enterprise Cloud
Built on enterprise-grade cloud infrastructure designed for scalability and reliability.
Ready to see AI-augmented clinical data review in action?
Start a Guided Trial
Experience Signals Clinical in a structured evaluation environment designed for real clinical review workflows. See how AI-assisted review accelerates listings, queries, and insights while maintaining traceability and oversight.
Explore a demo.
Watch a guided walkthrough of key capabilities. From listings and queries to Spotfire® visualizations.
Talk to an expert.
Discuss your data sources, timelines, and oversight requirements with a clinical analytics specialist.
Partner testimonial
Insights from Revvity Signals roundtable
At Revvity Signals Clinical roundtable, clinical, data, and medical leaders explored how AI can be responsibly applied to real-world clinical workflows. The discussion surfaced a clear consensus: progress depends on practical automation, human-in-the-loop governance, and trust-built systems that accelerate insight without compromising oversight or patient safety.
Resources







Signals Clinical FAQs
Ready to modernize clinical data review with Signals Clinical?
Start a Guided Trial
Experience Signals Clinical in a structured evaluation environment designed for real clinical review workflows. See how AI-assisted review accelerates listings, queries, and insights while maintaining traceability and oversight.
Explore a proof of concept
Talk with our team about a focused proof of concept tailored to your study workflows, data sources, and review needs.
Stay informed
Get updates on clinical oversight best practices, regulatory guidance, and product innovations from Signals Clinical.


























